  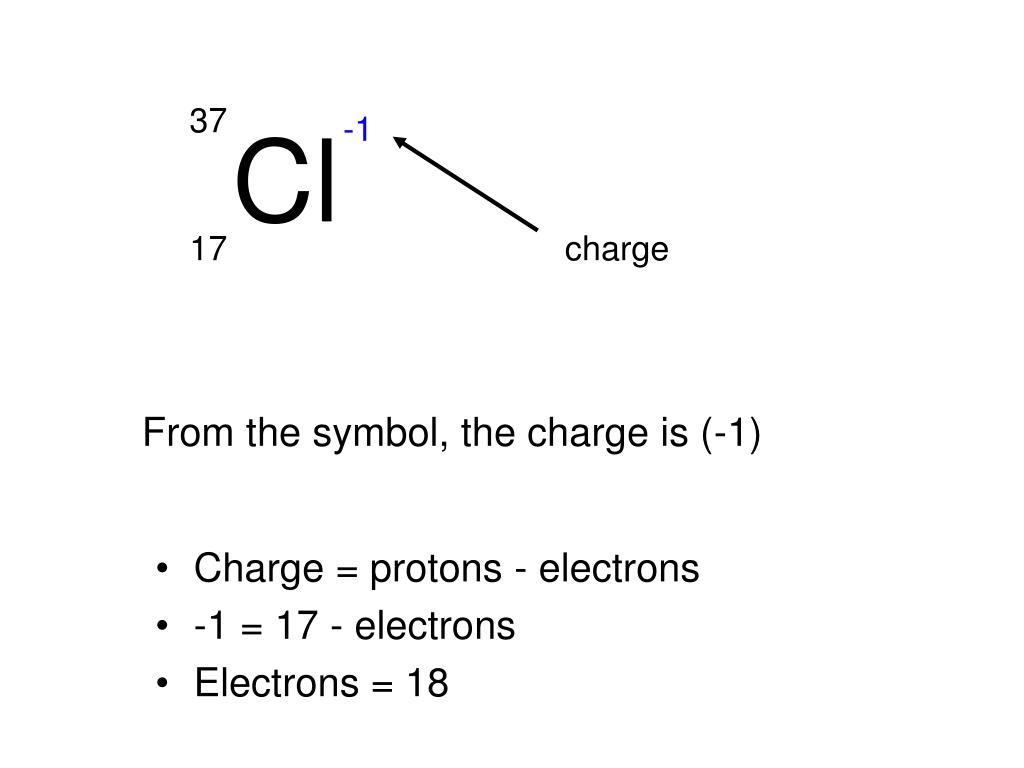
Negative and positive charges of equal magnitude cancel each other out. The particles found in nature allhave charges which are integral multiples of theelementary chargee: qnewheren. We have some articles in Universe Today that are related to the charge of the electron.\)) are useful, because, as you can see, the mass of a proton and the mass of a neutron are almost exactly \(1\) in this unit system. Thus, a basic Millikan Oil-Drop Experiment setup will include an enclosure containing falling charged oil drops, a device to measure their radii, an adjustable uniform electric field, and a meter to determine the field’s magnitude.īy repeating the experiment on a large number of oil drops, Millikan and his colleague, Harvey Fletcher, obtained electron charge values within 1% of the currently accepted one. Electrons are negatively charged particles and protons, of which nucleus is made of, are positively charged particles. is the mass number, symbol, and charge of an ion that contains 35 protons. 
Protons have a positive charge ( ) and electrons. magnitude of charge of electron e 1. Determine the number of protons, neutrons, and electrons in each element. The protons and electrons of an atom are attracted to each other. In SI units, the unit of charge is the coulomb (C). The charge on the ion tells you the number of. Answer:Option (A)Explanation:A body attains positive charge as some of the electrons removed from the body. At some point, when the upward force is equal to the downward force, the velocities may even go down to zero and the particles will stay in mid-air.Īt this specific instance, if we know the magnitude of the electric field (in N/C, units defining the force per unit charge) and the weight of each particle, we can calculate the force of the electric field on a single particle and finally derive the charge. magnitude F of the force between two charges q1 and q2 separated by a distance r is given by qq Fk r 12 2 where k constant 9.0 × 109 N m2/C2. However, it is possible to remove electrons and not change the identity of an element. The magnitude of the charge carried by a single electron or proton is usually considered to be the basic natural unit of charge and is given the symbol e. The lightest stable subatomic particle known, the electron carries a negative charge of 1. If the forces due to the field are directed opposite to gravity, the downward velocity of the particles may decrease. The elementary charge, usually denoted by e, is the electric charge carried by a single proton or, equivalently, the magnitude of the negative electric. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
